Acing the Tests
Experience is the best teacher for learning to spot contraband drugs and alcohol and drug intoxication in the field. But the cop needs a demonstrable standard for evidence. It's here that presumptive field testing for alcohol and drugs is most useful.
Experience is the best teacher for learning to spot contraband drugs and alcohol and drug intoxication in the field. Appearance, odor, context, packaging, and the reputation and demeanor of the people associated with the alcohol and/or drugs provide officers with more than enough information to develop what they believe to be probable cause for further investigation or an arrest.
Of course, "probable cause" isn't what a police officer believes to be sufficient evidence for an arrest - it's what a "reasonable person" would find sufficient. Those supposedly reasonable people don't see the world through a cop's eyes, so the cop needs a more demonstrable standard. It's here that presumptive field testing for alcohol and drugs is most useful.
TV and the movies used to show officers dipping their fingers into whatever powder they found, then tasting it before pronouncing it to be heroin or cocaine. The class on learning what drugs taste like wasn't included in my academy training, possibly because cyanide and cocaine look a lot alike. Media can be a more powerful teacher than formal training. In May 2010, a College Park (Ga.) PD police officer tested positive for cocaine after he backed his patrol car into another police vehicle. He claimed he taste-tested drugs he confiscated from suspects earlier in the day.
Presumptive drug testing is much safer, more defensible in court, and usually cheaper, if you include the cost of wrecked patrol cars and hospital bills for taste-testing cops. Most testing products marketed to law enforcement are chemical-based, self-contained, and disposable. Each test consumes a small sample of the drug and produces a color change for a positive result. There is another field testing method that consumes no sample at all, and indicates specifically what drug is present, e.g. opiate, cocaine, methamphetamine, etc.
Chemical Testing Kits
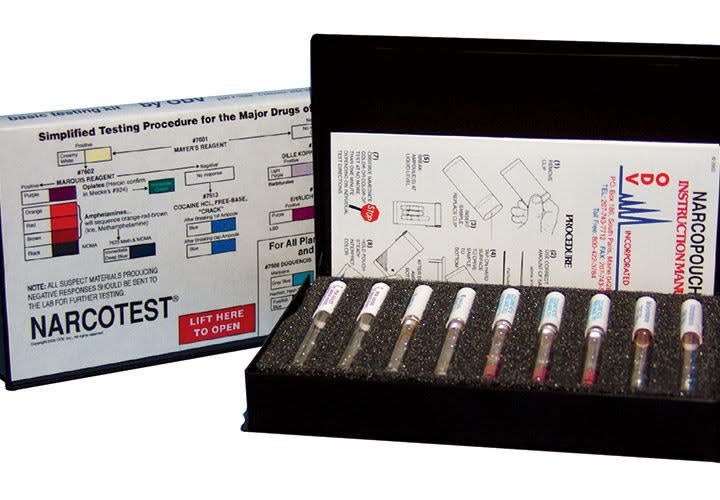
Self-contained chemical-based presumptive testing kits are available from companies like Jant Pharmacal (Accutest), BAE Systems (under the NIK and ODV labels), MMC International, and Mistral. The packaging differs, but the methodology is essentially the same.
The user inserts a small (pinhead-size) sample of the suspected drug into a plastic tube or chamber of the test unit, which is then sealed. The user then breaks a glass ampoule inside the tube or chamber, releasing the testing reagent, and shakes the tester to mix the sample and reagent. A specific color change indicates what drug, if any, is present in the sample. The user then books the self-contained tester into evidence, or more commonly, throws it away.
Variations of this process use multiple glass ampoules inside the test unit. If there is no color change after breaking the first ampoule, the user can break a second one inside the same kit to test for another substance.
A typical testing kit contains tubes or pouches for multiple drugs. Kits from Mistral have the user wipe a surface with a trace of the substance, using a special test paper. The user then sprays the paper with a reagent and looks for a color change.
Which vendor or product you choose or buy has a lot to do with the nature of the drug testing you do in the field. If you see mainly methamphetamine or cocaine on your stops, then it makes more sense to acquire test kits specific for that drug. A negative test won't tell you what drug (or baking soda) you have on hand, but you won't have a lot of unused tests going to waste, either.
If you deal with multiple drugs of abuse, and/or your officers aren't able to make an educated guess as to which drugs they have, then it's better to have a multi-drug test kit with a protocol for narrowing down the type of drug at hand. This can be an expensive approach. Each test tube or pouch costs from $1.25 to $7, depending on form factor, vendor, and drug type, and some protocols require up to seven tests before the user can say either "this is
Another factor to consider in your buying decision is shelf life and tolerance to environmental conditions. Most test kits have a shelf life from one to two years. Kits past their expiration date may yield spurious results, making their use worse than no test at all.
The reagents in the kits can be sensitive to temperature extremes and bright light. A kit carried in the trunk of a patrol car through a hot summer may undergo chemical changes to the reagents inside. A test conducted in freezing conditions might not be reliable, as most vendors recommend tests be conducted with everything at room temperature.
Each agency's situation is unique. Analyze the variables of common drugs encountered, sophistication of your officers, number of tests conducted, cost, temperature, and other environmental extremes so that you'll be prepared with the right questions for potential suppliers.[PAGEBREAK]
No-touch Analysis
A relatively new application of an old technology allows officers to identify suspect drugs without sampling or touching the drug, and sometimes without even opening its packaging.
Raman spectroscopy (named after one of the people who first described it in 1928) measures the shift in energy of photons emitted when laser light strikes the substance in question. This energy profile is unique for each molecule, so any chemical substance is identifiable by comparing the profile against a standard. There's no need to sample or touch the substance in question. The analysis can be done through a plastic bag or other clear container.
Full-scale Raman spectroscopy requires quite a bit of equipment, but the bulk is reduced considerably when you limit the substances you can identify. A device called the ReporteR packs the technology into a handheld device a bit larger than a pack of cigarettes. The device, manufactured by DeltaNu, contains the laser, a collector for the emitted photons, and a database of profiles of commonly encountered drugs of abuse.
When a user points the laser at a suspect drug, the device analyzes the profile of the substance and compares it to the information stored in the ReporteR. A display shows a match with one of the stored profiles or a "no match" indication. There is enough memory to hold the profiles of nearly all commonly encountered drugs of abuse, so this is a single-test solution with no consumables. The device comes with a liquid vial attachment for analysis of solutions. The operator can save each test result and print out a report later, if desired.
Alcohol Testing in the Field
Although the number of alcohol-related fatalities has steadily declined, driving under the influence of alcohol remains a major factor in injury and non-injury accidents, and an enforcement priority for most police agencies.
Field sobriety testing is more standardized and definitive than ever before. Still, there is an experiential factor for officers administering these tests, and defense attorneys will use any actual or perceived error to try to discredit the officer and his or her decision to arrest. Fortunately, there is technology to aid officers in taking drunk drivers off the road.
Breath testing is the most common method for measuring blood-alcohol concentration (BAC). While not as accurate as a blood test, it is less invasive, faster, and less expensive than drawing blood and having it analyzed in a laboratory. Breath testing technology has been slowly evolving for more than 70 years, and nearly every method is still used today in some form.
The "drunkometer" was first used in 1938. The subject blew into a tube that inflated a balloon inside the test device. The breath sample was pumped through an acidified solution of potassium permanganate, which changed color if alcohol was present. The greater the color change, the more alcohol was present.
Today, companies such as Dräger (which also produces sophisticated evidentiary breath testers and other medical equipment) still sells Alcotest Tubes, which use a similar chemical reaction color change to show the presence of alcohol in a breath sample.[PAGEBREAK]
Evidentiary Breath Testers
Most evidentiary breath testers in use today employ infrared spectroscopy to measure BAC. The operating principle of infrared spectroscopy or spectrophotometry is that every substance will absorb infrared light at wavelengths unique to that substance. When a breath sample is introduced into the sample chamber of a breath analyzer, infrared light is beamed through the transparent chamber toward a photocell on the opposite side.
Any alcohol present in the breath sample will absorb the relevant wavelengths of light, and the more alcohol present in the sample, the more light is absorbed. The analysis is specific for ethanol, so other volatiles in the sample (such as acetone, produced by diabetics and some dieters) that contributed to falsely high readings in more primitive devices have no effect.
Some accused drunk drivers will claim their BAC reading came from mouth or stomach alcohol. A denture or other mouth structure or the stomach gas coming from a burp can contain alcohol. Pre-test procedures of rinsing the mouth with water and observing the subject for burping are supposed to prevent these claims, but people still try.
Fact is, both mouth and stomach sources, if measured as lung air, will show lethal BAC levels. Either one will begin to dissipate with exhalation, so the BAC level in the sample will decrease as the subject empties his or her lungs. Modern testers include "slope indicators" that refuse a sample where the BAC level decreases as it passes through the sensor.
Handheld Breath Testers
Preliminary breath testers (PBTs) used by police in the field employ platinum fuel cells to measure BAC. Breath samples pass through electrodes coated with a thin layer of platinum black. Any alcohol present in the sample is oxidized, releasing electrons. The free electrons pass from one electrode to the other, creating a current. The current is proportional to the amount of alcohol in the breath sample.
PBTs don't have slope detectors, and they can be fooled by some other organic compounds. They are reliable enough for field work, although most states do not allow their results to be introduced in court as evidence. Their main function is to assist the officer in establishing probable cause for the arrest.
There is a fourth technology for measuring BAC, used primarily in consumer-grade electronic breath testers. Semiconductor-based devices can detect alcohol in a breath sample, but their measurements of BAC are unreliable. They are prone to false positives, showing a BAC when there is no alcohol present.
They are also prone to contamination, and the sensors require recalibration or replacement every six months to preserve whatever accuracy they do have. Few consumers will bother to do this. No state allows the results from a semiconductor-based tester to be used as evidence of BAC, and the U.S. Deptartment of Transportation rejects them as well.
You have many options for performing field tests of drugs and impaired drivers. Understanding the technology behind them will help you make better decisions on which ones to use.
Tim Dees is a retired police officer and the former editor of two major law enforcement Websites. He can be reached at editor@policemag.com.
DRUG AND ALCOHOL TESTS
Accutest/Jant Pharmacal
Acunetx
Ahura Scientific
Alcohol Countermeasure Systems
CMI, Inc.
DeltaNu
Drägerwerk AG & Co.
Icx Technologies
Mistral Group
MMC International
Morphix Technologies
PAS Systems International
Safariland
More Patrol

Why Tennessee’s New Deadly Force Law Matters Beyond Tennessee
Tennessee’s new deadly force law is more limited than many may realize. Effective July 1, 2026, the law applies to a person who is not engaged in conduct that would constitute a felony or a Class A misdemeanor and who is in a place where the person lawfully resides. But it could have far-reaching influence.
Read More →
When Do You Transport a Wounded Officer by Police Vehicle?
Time-sensitive injuries like penetrating trauma mean a wounded officer needs to reach a trauma center as soon as possible, and in some cases, that means transporting by police vehicle rather than waiting for EMS. What are the factors to consider in making that transport decision?
Read More →
Safariland Solis Rethinks Concealable Duty
What if Level I retention didn’t require a full duty rig? Safariland’s Solis delivers trusted ALS security in a streamlined OWB platform built for administrative and plainclothes professionals who need protection without the bulk.
Read More →
5 Things to Know When Buying Backup Lights for Patrol Use
What is the value of a backup light, and what do you need to consider when selecting one? These smaller lights are invaluable as a secondary or special-purpose light and can be easily carried in a pocket or clipped to MOLLE gear, a key chain, shirt, or a vest.
Read More →
363 Fallen Officers Honored During National Police Week
In case you missed NELOMF’s annual Candlelight Vigil, here are all 363 names of the fallen officers whose names were added to the National Law Enforcement Officers Memorial this year.
Read More →
NYPD Officers Who Responded Quickly During IED Incident Recognized as NLEOMF Officers of the Month
Chief Aaron Edwards and Sgt. Luis Navarro were recognized by NLEOMF as Officers of the Month for their response during an IED incident and their actions that helped prevent harm to the public.
Read More →
National Police Survey Reveals How Americans View Policing Today
A new national survey offers a comprehensive look at how Americans view policing. The study uncovers public sentiment toward local vs. federal police, communications, crisis response, the use of AI in policing, and more.
Read More →
Streamlight TLR-7 X Selected as Standard Issue Pistol Light by Canadian Federal Police
The Canadian Federal Police will be adding a new duty pistol system, which will include Streamlight’s TLR-7 X and a Glock 45 MOS 7 pistol with a red dot sight.
Read More →
Fallen Law Enforcement Officers from Across the Country to be Honored During 38th Annual Candlelight Vigil on May 13th in Washington, D.C.
The National Law Enforcement Officers Memorial Fund (NLEOMF) will honor 363 fallen officers who have died in the line of duty as their names are added to the National Law Enforcement Officers Memorial during the annual Candlelight Vigil on May 13.
Read More →
What Should Be in Your IFAK?
What should every officer include in an IFAK? Sydney Vail, M.D., a veteran trauma surgeon and former SWAT surgeon, explains which components are needed and which are not, and stresses training.
Read More →

